A study to find the utility of ZN staining in the diagnosis of confirmed cases of bone tuberculosis
Abstract
Introduction: As per the WHO guidelines, confirmed and definitive microbiological diagnosis of OATB is required before initiation of anti TB treatment. A study was conducted to find the utility of ZN staining in the diagnosis of confirmed cases of OATB cases.
Materials and Methods: Study was conducted in the Department of Microbiology, GSL Medical College. Informed written consent was taken from all the volunteers. The study protocol was approved by the institutional ethics committee. Individuals aged > 18 years who were clinically confirmed to be OATB were included in the study. Individuals with acute pyogenic osteoarticular infections were excluded. A simple FNAC technique was conducted in the outpatient unit for the collection of aspiration. Smears were prepared and stained by Ziehl Neelsen staining. Smear preparation and staining were done as per the RNTC guidelines.
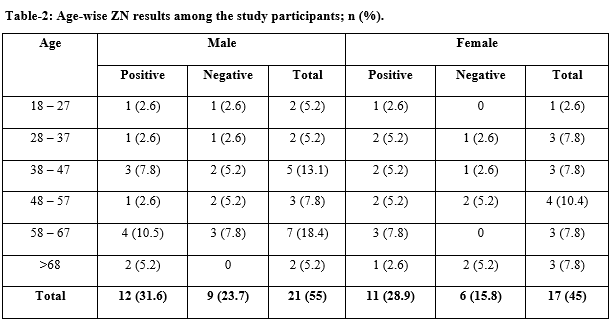
Results: During the study period 38 participants with suspected OATB were included. The smear positivity was 23 (60.5%). The male-female ratio was 1.08; statistically there was no significant difference (P = 0.753954). Age-wise maximum smear-positive cases were diagnosed in 58-67 years age group.
Conclusion: In resource-limited areas, ZN staining is a good alternative for the diagnosis of OATB.
Downloads
References
Merino P, Candel FJ, Gestoso I, Baos E, Picazo J. Microbiological diagnosis of spinal tuberculosis. Int Orthopaed. 2012;36(2):233–238. doi: https://dx.doi.org/10.1007%2Fs00264-011-1461-x.
WHO (2009). Annual report on Global tuberculosis control: epidemiology, strategy, financing. Geneva: World Health Organization, 2009. Available at https://www.who.int/tb/publications/global_report/en/.
Sachdeva KS, Ashok K, Dewan P, Ajay K, Satyanarayana S. New Vision for Revised National Tuberculosis Control Programme (RNTCP): Universal access-Reaching the unreached. Ind Med Res. 2012;135(5):690-694.
Sitanshu SK, Archana Ramalingam. Same day sputum microscopy: The road ahead in tuberculosis diagnosis. Lung India. 2013;30(3):226-227. doi: https://doi.org/10.4103/0970-2113.116242.
Agarwal A, Singh S, Agarwal S, Gupta S. Needle aspiration and cytology for suspected osteoarticular tuberculosis in children. Malaysian Ortho J. 2018;12(3):31-37. doi: https://dx.doi.org/10.5704%2FMOJ.1811.007.
Shah MM, Jhaveri SN, Tuli SM. Musculoskeletal Tuberculosis in Children. Surgery in Africa. 2011:1 – 12.
Chandra T J. Same day sputum smear microscopy approach for the diagnosis of pulmonary tuberculosis in a microscopy center at Rajahmundry. Indian J Tuberc 2012; 59: 141–144.
Chandra TJ, Raj R S, Sharma Y V. Same day sputum smear microscopy approach with modified ZN staining for the diagnosis of pulmonary tuberculosis in a microscopy centre at Rajahmundry. Indian J Med Microbiol. 2014;32(2):153-156. doi: https://doi.org/10.4103/0255-0857.129801.
RNTCP Central TB Division. Manual for Laboratory Technicians. New Delhi, India: Directorate General of Health Services, Ministry of Health and Family Welfare, 1998. Available from: http://www.tbcindia.org/LABMANUAL.pdf [Last accessed date on 2018 Jan 22].
Chandra TJ. One-sample two-smear versus two-sample two-smear approach for the diagnosis of pulmonary tuberculosis. J Lab Physicians. 2018;10(2):135-139. doi: https://dx.doi.org/10.4103%2FJLP.JLP_145_17.
Chandra TJ, Raj RS, Sharma YV. Same day sputum smear microscopy for the diagnosis of pulmonary tuberculosis: Direct versus concentrated sputum smear. Int J Tuberc Lung Dis. 2016;20(2):247-251. doi: https://doi.org/10.5588/ijtld.15.0566.
Arathi N, Ahmad F, Huda N. Osteoarticular tuberculosis – a three years’ retrospective study. J Clin Diagn Res. 2013;7(10):2189-2192. doi: https://dx.doi.org/10.7860%2FJCDR%2F2013%2F6859.3467.
Masood S. Diagnosis of tuberculosis of bone and soft tissue by fine-needle aspiration biopsy. Diagn Cytopathol. 1992;8(5):451-455. doi: https://doi.org/10.1002/dc.2840080505.
Mousa HA. Tuberculosis of bones and joints: diagnostic approaches. Int Orthop. 1998;22(4):245-246. doi: https://dx.doi.org/10.1007%2Fs002640050251.
Beiner JM, Grauer J, Kwon BK, Vaccaro AR. Postoperative wound infections of the spine. Neurosurg Focus. 2003;115(3):E14. doi: https://doi.org/10.3171/foc.2003.15.3.14.
Sedick Q, Vaughan J, Pheeha T, Alli NA. Bone marrow aspirate microscopy v. bone marrow trephine biopsy microscopy for detection of Mycobacterium tuberculosis infection. South Amer Med J. 2015;105(9):773-776. doi: https://doi.org/10.7196/samjnew.8171.
Cuevas L E, Yassin M A, Al-Sonboli N, et al. A multi-country non-inferiority cluster randomized trial of frontloaded smear microscopy for the diagnosis of pulmonary tuberculosis. PLOS Med. 2011;8:e1000443. doi: https://doi.org/10.1371/journal.pmed.1000443.
Chandra T J, Alam R R, Selvaraj R, Sharma Y V. MODS assay for rapid diagnosis of Tuberculosis among HIV TB co infected individuals in a tertiary care hospital, Andhra Pradesh. Pak J Chest Med. 2014;20(4):133-138.
Hooja S, Pal N, Malhotra B, Goyal S, Kumar V, Vyas L. Comparison of ZiehlNeelsen& Auramine O staining methods on direct and concentrated smears in clinical specimens. Indian J Tuberc. 2011;58(2):72-76.


 OAI - Open Archives Initiative
OAI - Open Archives Initiative


















 Therapoid
Therapoid

